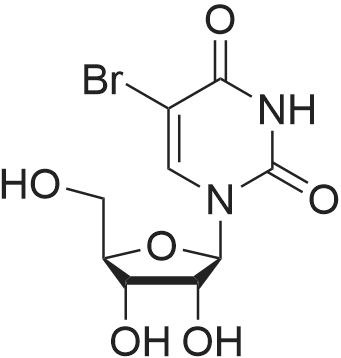
| CAS No. : | 957-75-5 | MDL No. : | MFCD00006528 |
| Formula : | C9H11BrN2O6 | Boiling Point : | - |
| Linear Structure Formula : | - | InChI Key : | AGFIRQJZCNVMCW-UAKXSSHOSA-N |
| M.W : | 323.10 | Pubchem ID : | 91494 |
| Synonyms : |
BrU;NSC 38296;bromouridine;Uridine, 5-bromo-;957-75-5;BrUrd;(–)-5-Bromouridine
|
Chemical Name : | 5-Bromo-1-((2R,3R,4S,5R)-3,4-dihydroxy-5-(hydroxymethyl)tetrahydrofuran-2-yl)pyrimidine-2,4(1H,3H)-dione |
| Signal Word: | Warning | Class: | N/A |
| Precautionary Statements: | P261-P305+P351+P338 | UN#: | N/A |
| Hazard Statements: | H302-H315-H319-H335 | Packing Group: | N/A |
| GHS Pictogram: |

|
||